The mass flux of magmatic gases from Mt Merapi has been demonstrated by Allard et al. (1995) to exceed lava effusion rates, indicating degassing of un-erupted lava in the magmatic system. This degassing is estimated to be 40 times greater than the lava effusion rate. Clues to the make up of Merapi's magmatic system are therefore provided through analyses of gases and fluids in fumaroles near the volcanoes summit.
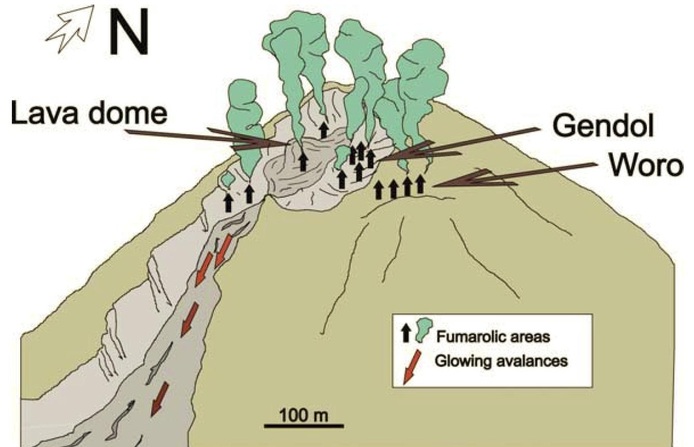
Figure 22: Schematic of the upper slopes of My Merapi depicting the lava dome and locations of fumaroles. From Zimmer and Erzinger (2003).
Geothermal system
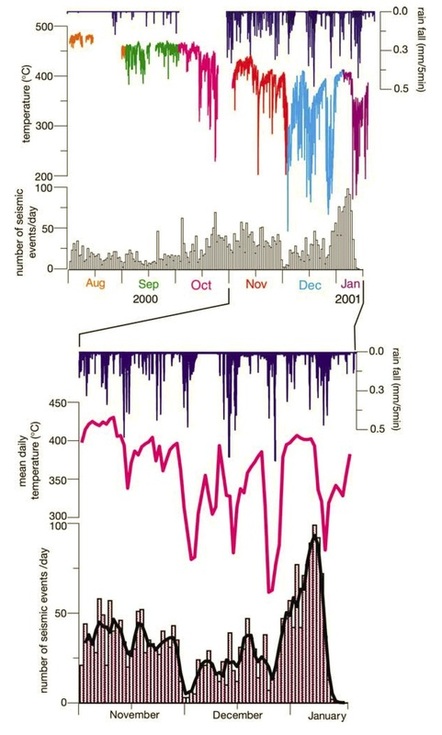
Two high temperature hydrothermal fields are recognized on Merapi: Gendol and Woro (Figure 22). These are located 150 and 250 m from the summit on the SE side of the volcano. Temperatures of the fumaroles at Gendol are as high as 800 degrees C, whereas at Woro temperatures can be as high as 600 degrees C. Zimmer and Erzinger (2003) recorded a unique relationship between the hydrothermal system, rainfall, and seismicity on the volcano (Figure 23). Periods of high rainfall are observed to coincide with reduced temperatures in the hydrothermal system and a corresponding reduction in the amount of seismicity.
Gas mixtures at fumaroles are primarily composed of H2O (84-92%) and CO2 (8-16%) (Zimmer and Erzinger, 2003). SO2 occurs in trace amounts, but rises significantly before and during eruptions. Periodic increases of CO2 concentrations are thought to be the result of degassing magma.
Figure 23: Comparison plots of rainfall, temperature of the hydrothermal system, and seismic events at various times from 2000-2001. The lower plot highlights trends from November 2000 to January 2001. Note the correlation between peaks in rainfall (blue histogram) and mean temperature (pink graph). These in turn appear to have an effect on the number of seismic events per day (black lines). Graphs from Zimmer and Erzinger (2003).
Gas mixtures at fumaroles are primarily composed of H2O (84-92%) and CO2 (8-16%) (Zimmer and Erzinger, 2003). SO2 occurs in trace amounts, but rises significantly before and during eruptions. Periodic increases of CO2 concentrations are thought to be the result of degassing magma.
Figure 23: Comparison plots of rainfall, temperature of the hydrothermal system, and seismic events at various times from 2000-2001. The lower plot highlights trends from November 2000 to January 2001. Note the correlation between peaks in rainfall (blue histogram) and mean temperature (pink graph). These in turn appear to have an effect on the number of seismic events per day (black lines). Graphs from Zimmer and Erzinger (2003).
Whole-rock geochemistry
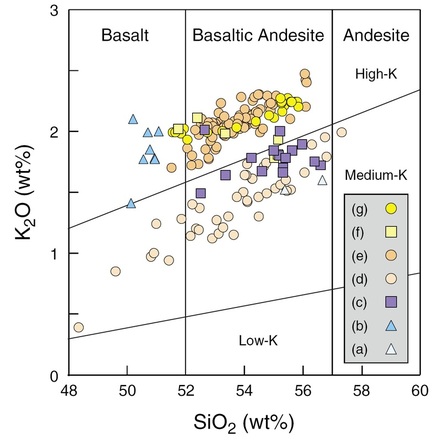
Whole-rock geochemical analyses of Mount Merapi rocks reveal deposits that vary from calc-alkaline basalts to basaltic andesites with medium to high potassium contents (Gertisser et al., 2012: Figure 24). Lavas erupted from Merapi are, however, generally basaltic andesite in composition.
A shift in whole-rock chemistry is observed at ~1,900 yrs BP, where a higher proportion of high-K eruptive products are observed (Gertisser et al., 2012). This change in chemistry roughly coincides with the formation of the New Merapi cone, which occurred somewhere between 4.8 + 1.5 ka and 1.59 + 0.04 ka.
Figure 24: K2O vs SiO2 classification diagram for Mt Merapi rocks from Gertisser et al. (2012).
Phases of high eruption frequency at Mount Merapi, occurring most recently at 1,300-1,150 and 700-550 yrs BP, are characterized by SiO2-rich whole-rock compositions during the earliest phases of activity (Gertisser and Keller, 2003). The high silica contents observed at these early stages are thought to result from differentiation processes occurring during the preceding period of reduced activity. As these periods of high eruption frequency proceed, less evolved compositions are eventually erupted. Individual, large eruptions (VEI 3-4), however, exhibit wide variations in SiO2 (e.g. 51.5-57.2%), which Andreastuti et al. (2000) attribute to recharge events from a deeper chamber.
A shift in whole-rock chemistry is observed at ~1,900 yrs BP, where a higher proportion of high-K eruptive products are observed (Gertisser et al., 2012). This change in chemistry roughly coincides with the formation of the New Merapi cone, which occurred somewhere between 4.8 + 1.5 ka and 1.59 + 0.04 ka.
Figure 24: K2O vs SiO2 classification diagram for Mt Merapi rocks from Gertisser et al. (2012).
Phases of high eruption frequency at Mount Merapi, occurring most recently at 1,300-1,150 and 700-550 yrs BP, are characterized by SiO2-rich whole-rock compositions during the earliest phases of activity (Gertisser and Keller, 2003). The high silica contents observed at these early stages are thought to result from differentiation processes occurring during the preceding period of reduced activity. As these periods of high eruption frequency proceed, less evolved compositions are eventually erupted. Individual, large eruptions (VEI 3-4), however, exhibit wide variations in SiO2 (e.g. 51.5-57.2%), which Andreastuti et al. (2000) attribute to recharge events from a deeper chamber.
Magma-carbonate interactions
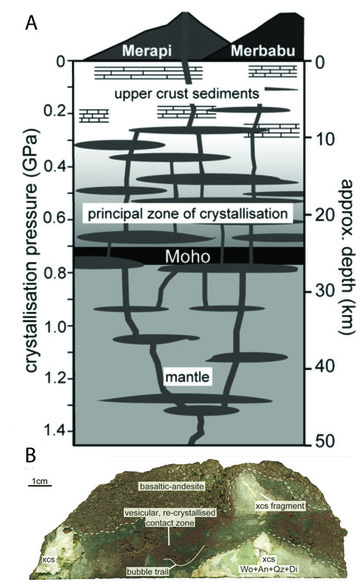
Samples of Merapi's volcanic rock reveal evidence for the interaction between uppercrustal carbonate rocks (limestone) and magma prior to eruptions (Deegan et al., 2010; 2011; Figure 25). The assimilation of limestone into rising magma results in CO2 bubble growth, and with further bubble expansion the intensity of eruptions will increase markedly. Breaking of country rock during large earthquakes is also expected to change the nature of fracture networks within the limestone, and potentially enhance the movement CO2 in the crust (Deegan et al., 2011). It follows that earthquake events at Merapi have the potential to greatly alter the chemistry and volatile content of the magma system, possibly triggering eruptions or leading to enhanced activity, as observed in May 2006 immediately after the magnitude 6.3 Yogyakarta earthquake.
Figure 25: (A) Schematic illustration of the Mt Merapi magmatic system, showing ~10km of upper-crustal carbonate rocks. (B) Image of a Mt Merapi lava rich in calcareous, crustal xenoliths (xcs). From Deegan et al. (2010).
